Blog post by Dr. Tamara Maiuri
Another quick report on some year-old data that missed the boat on Zenodo deposits.
How to measure huntingtin chromatin retention
The method for measuring huntingtin chromatin retention upon oxidative stress makes use of a fluorescent huntingtin-specific intrabody, so we can follow the movement of endogenous huntingtin.
The method is described in this Zenodo post, including all the necessary controls to make sure the huntingtin chromatin retention is specific to huntingtin, and specific to oxidative stress conditions. Briefly, the YFP-tagged huntingtin-specific intrabody is co-transfected with H2B-mCherry (transfection control and marker of chromatin). After treatment, soluble proteins are extracted and cells are fixed and imaged. CellProfiler [1] is used to identify nuclei by the H2B-mCherry signal, and the nuclear YFP signal remaining bound to chromatin is quantified for several hundred cells.
Is PARP activity required for huntingtin chromatin retention?
Once these conditions were established, I was able to apply them to ask a biological question: is PARP activity required for huntingtin chromatin retention?
The Keith Caldecott lab at University of Sussex kindly provided us with a panel of PARP knock out cell lines. These are RPE1 cells with either PARP1 or PARP2 or both PARP1/PARP2 deleted by CRISPR/Cas9 [2].
When I first did this experiment, I was somewhat confused by the results. Huntingtin chromatin retention was diminished in PARP1 knockout cells and in PARP2 knockout cells, suggesting a role for PAR in the recruitment of huntingtin to chromatin. This would be expected based on our previous results that huntingtin binds PAR and acts as a scaffold for DNA repair proteins [3].

However, the double knockout cells seemed to retain huntingtin at chromatin more efficiently than either single knockout cell line. This didn’t make sense until I noticed that the huntingtin intrabody expression pattern in the double knockout cells had very bright nuclear and cytoplasmic staining (almost like inclusions) compared to the diffuse nuclear staining seen in wild type cells. To take a closer look, I compared the expression patterns by high magnification microscopy.
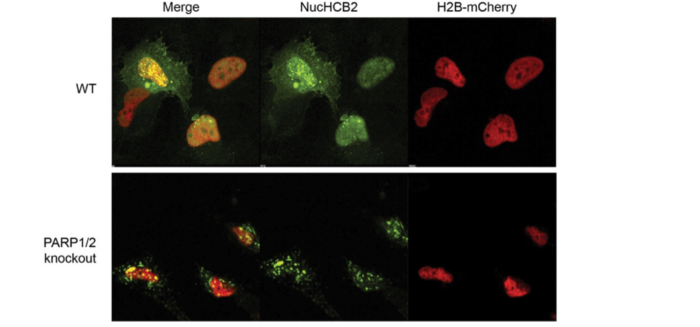
In wild type RPE1 cells, the intrabody is recruited to the chromatin in response to KBrO3 and displays diffuse nuclear localization. A small proportion of cells also have bright punctate staining in the nucleus and cytoplasm. In PARP1/2 double knockout cells, on the other hand, the intrabody forms large inclusions, some of which may be positioned over the nucleus. If so, in the quantification process, these inclusions would be scored as nuclear nucHCB2 intensity within the confines of the H2B-mCherry-delineated nucleus, skewing the results.
So I acquired a Z-stack to see whether the inclusions are within or above the nucleus:

Sure enough, the bright inclusions are positioned above the nucleus. This means that huntingtin localization is somehow dysregulated in PARP1/2 double knock out cells. Cytoplasmic inclusion formation skews CellProfiler results when inclusions are positioned over the nucleus. Transient transfection of the huntingtin-specific intrabody is therefore not the method of choice for measuring huntingtin chromatin retention in these cells.
The experimental details and results of huntingtin chromatin retention for all three cell lines have now been deposited to Zenodo.
In the end, what we have are a few more clues. The fact that huntingtin chromatin retention is diminished in PARP1 KO cells and PARP2 KO cells is worth investigating further. The fact that huntingtin localization is dysregulated in the double KO cells is also worth pursuing, but it makes these cells unusable for further experiments of this type.
- Carpenter AE, Jones TR, Lamprecht MR, Clarke C, Kang IH, Friman O, et al. CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 2006;7: R100.
- Hanzlikova H, Gittens W, Krejcikova K, Zeng Z, Caldecott KW. Overlapping roles for PARP1 and PARP2 in the recruitment of endogenous XRCC1 and PNKP into oxidized chromatin. Nucleic Acids Res. 2017;45: 2546–2557.
- Maiuri T, Mocle AJ, Hung CL, Xia J, van Roon-Mom WMC, Truant R. Huntingtin is a scaffolding protein in the ATM oxidative DNA damage response complex. Hum Mol Genet. 2017;26: 395–406.
